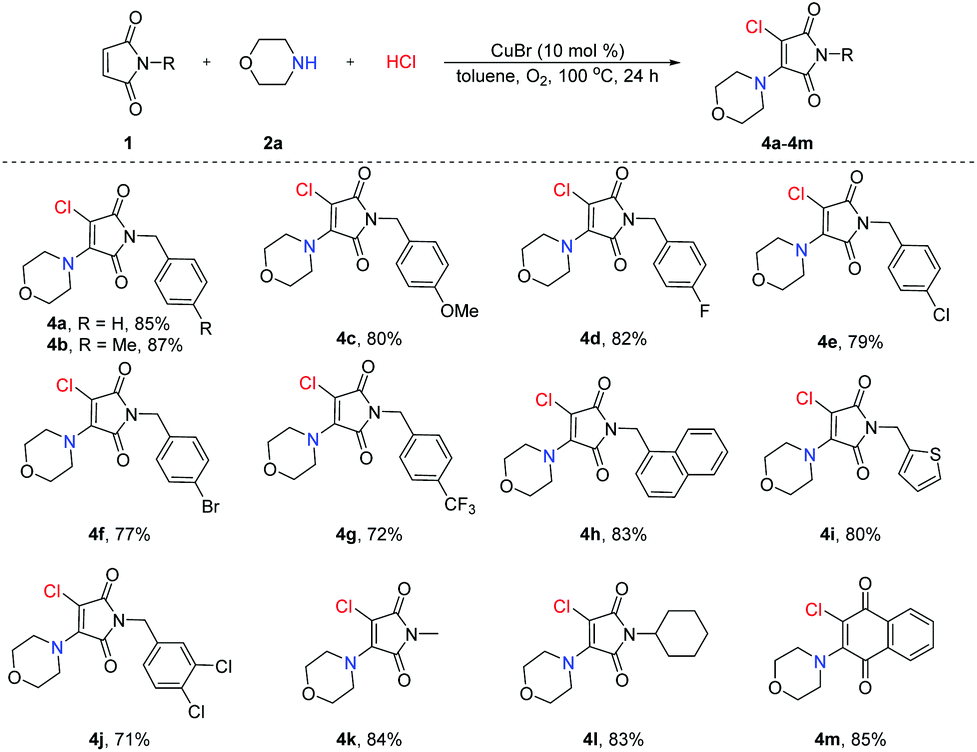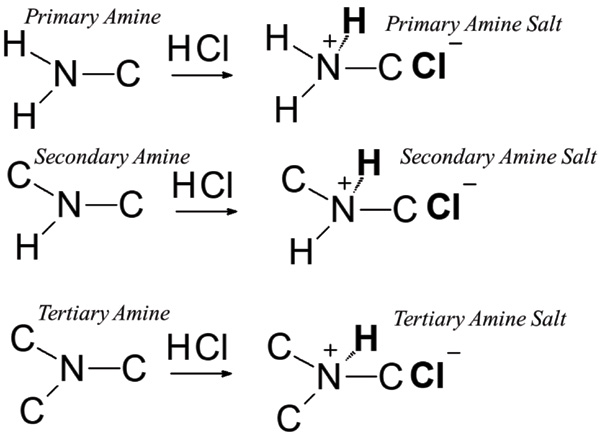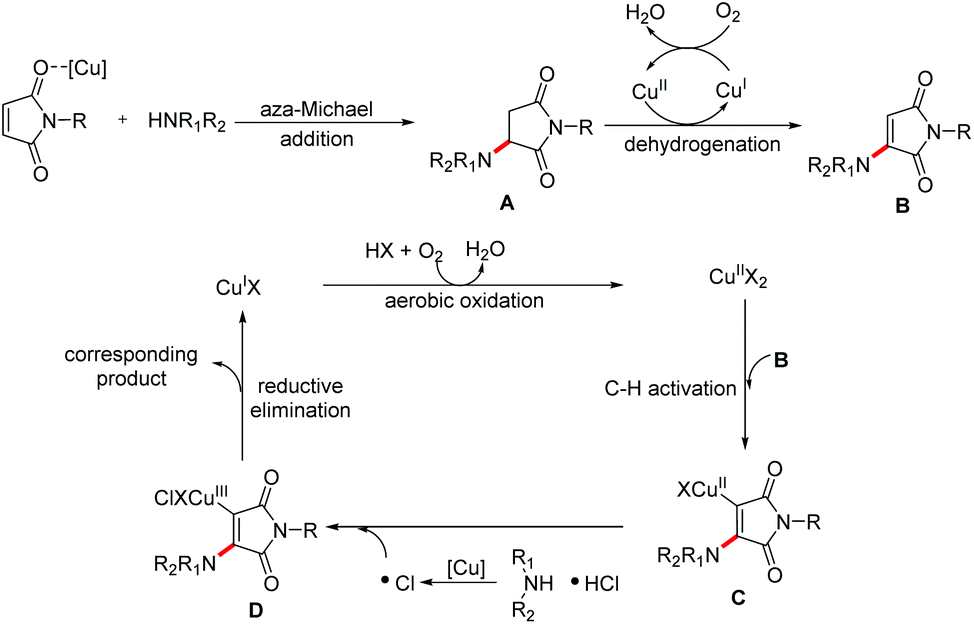
Amine hydrochloride salts as bifunctional reagents for the radical aminochlorination of maleimides - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00916H
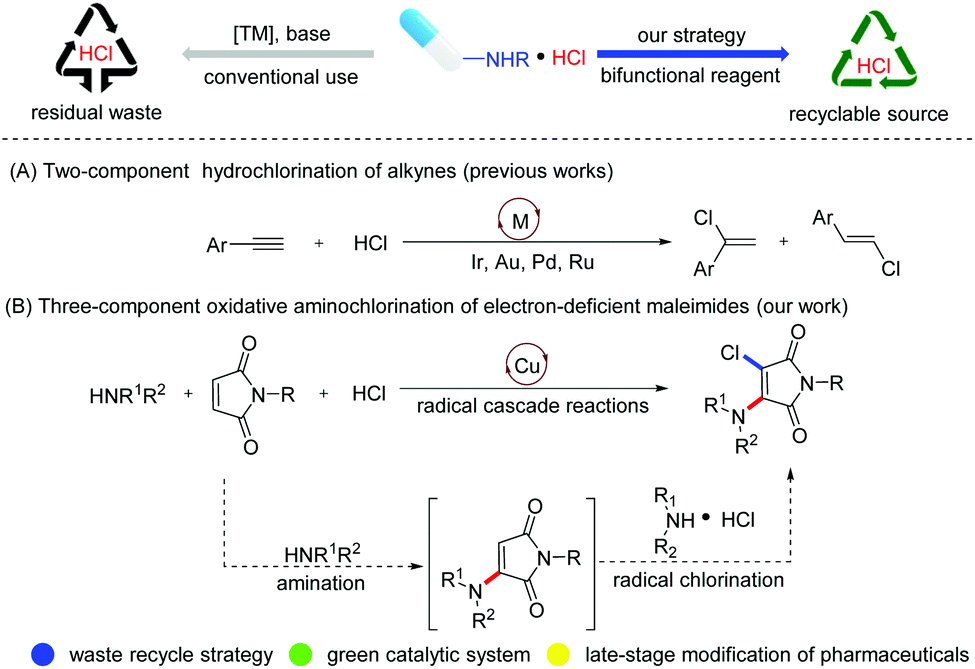
Amine hydrochloride salts as bifunctional reagents for the radical aminochlorination of maleimides - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/D1QO00916H

Write a chemical equation for the conversion of the following carboxylic acid salt to its parent carboxylic acid. Let hydrochloric acid (HCl) be the source of the needed hydronium ions. Sodium lactate.